It is therefore well suited for binding studies where de novo assignments of amide 1 H and 15 N resonances need to be obtained, even in cases where sensitivity is the limiting factor.īEST-TROSY cell-free expression cyclophilin D ligand screening selective isotope labeling. This scheme was combined with time-shared triple-resonance NMR experiments, which allows a fast and efficient backbone assignment by giving the unambiguous assignment of unique amino acid pairs in the protein, the identity of ambiguous pairs and information about all 19 non-proline amino acid types. Using a cell-free expression system, a scheme that involves 15 N, 1- 13 C, 2- 13 C, fully 15 N/ 13 C, and unlabeled amino acids was optimized to gain a maximum of assignment information from three samples. They are structurally and chemically different, and also differ in size and volume. In this study, combinatorial 15 N-, 13 C α -, and 13 C'-selective labeling has been used during the backbone assignment of human cyclophilin D to explore binding of an inhibitor molecule. They are composed of C, H, O, N and S atoms. Its the side chain that is specific to each individual type. 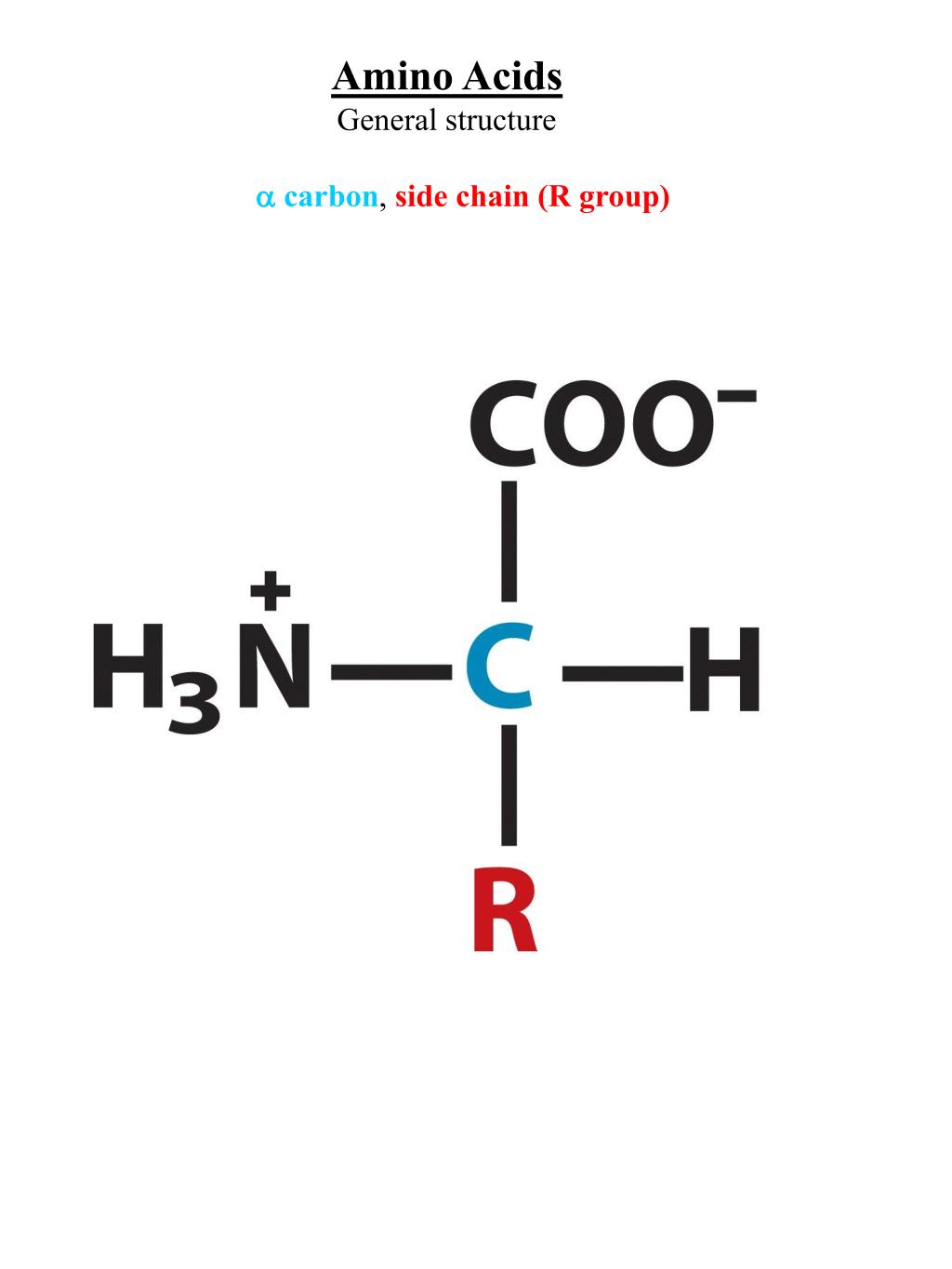
This backbone is the same in all amino acids. Nucleic Acid nucleosides, nucleotides, RNA, and DNA Residues residues in currently open structures, sorted into nonstandard residues, standard nucleic acids, and standard amino acids Structure. The modification occurs after the amino acid has been assembled into a protein.Selective labeling with stable isotopes has long been recognized as a valuable tool in protein NMR to alleviate signal overlap and sensitivity limitations. The amino and carboxyl groups and central carbon are considered the amino acid backbone. In some cases an amino acid found in a protein is actually a derivative of one of the common 20 amino acids (one such derivative is hydroxyproline). Glycine, the major amino acid found in gelatin, was named for its sweet taste (Greek glykys, meaning “sweet”). It was obtained from protein found in asparagus juice (hence the name). The first amino acid to be isolated was asparagine in 1806. The only amino acid whose R group has a pK a (6.0) near physiological pHĪlmost as strong a base as sodium hydroxide 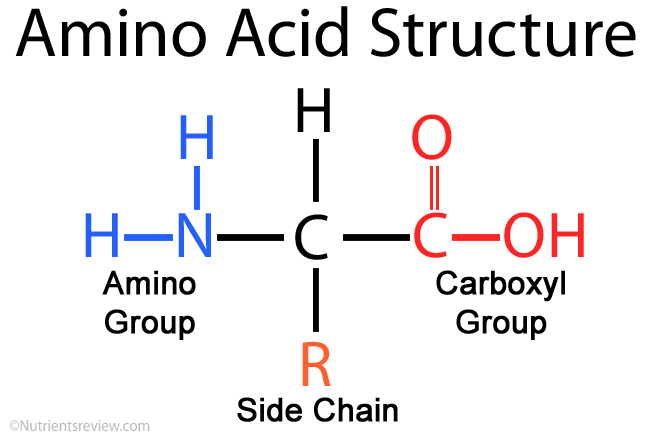
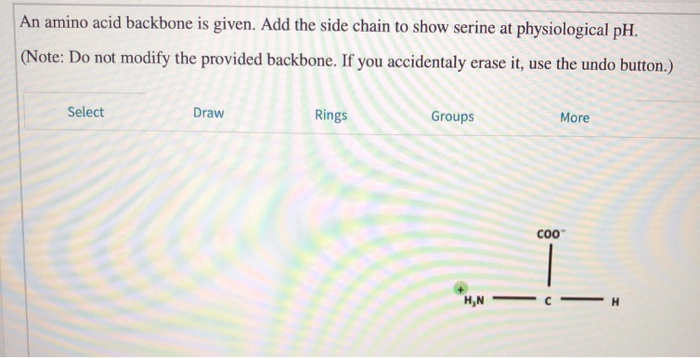
An amino group is a nitrogen atom that is single bound to two or three. Nonpolar amino acids (there are 9) contain aliphatic (hydrocarbon) chains or aromatic rings. Oxidation of two cysteine molecules yields cystineĪmino acids with a negatively charged R groupĬarboxyl groups are ionized at physiological pH also known as aspartateĬarboxyl groups are ionized at physiological pH also known as glutamateĪmino acids with a positively charged R group The amino acid backbone consists of an amino group that is bound to a central carbon that is bound to a carboxyl group. Named for its similarity to the sugar threose Side chain functions as a methyl group donorĬontains a secondary amine group referred to as an α-imino acidĪmino acids with a polar but neutral R group The 'R' group varies among amino acids and determines. All amino acids have the alpha carbon bonded to a hydrogen atom, carboxyl group, and amino group. 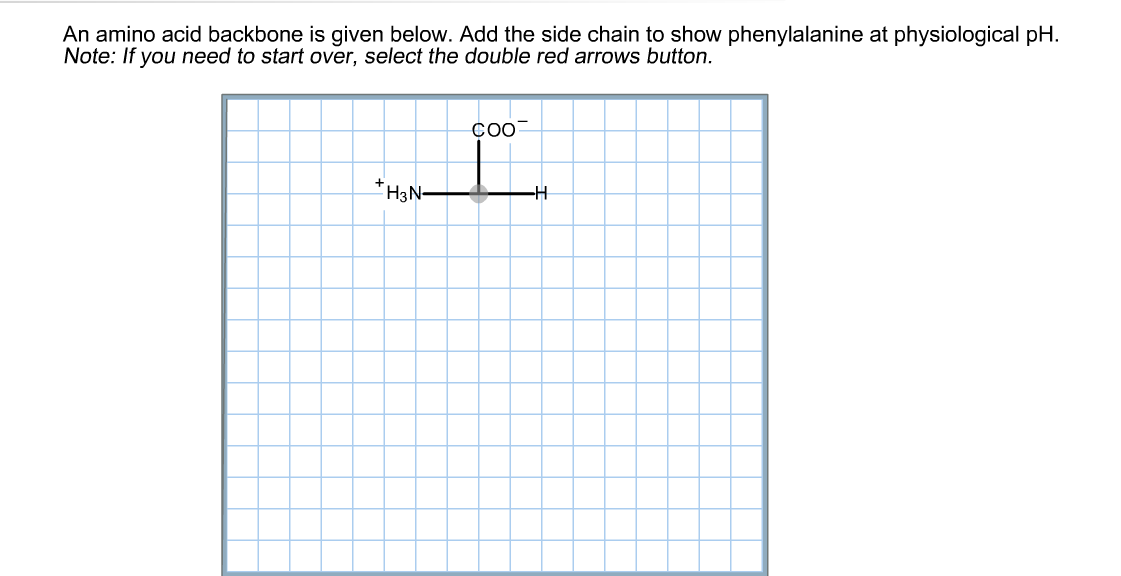
The only amino acid lacking a chiral carbonĪn essential amino acid because most animals cannot synthesize branched-chain amino acidsĪlso classified as an aromatic amino acid Generally, amino acids have the following structural properties: A carbon (the alpha carbon) A hydrogen atom (H) A Carboxyl group (-COOH) An Amino group (-NH 2) A 'variable' group or 'R' group. \): Common Amino Acids Found in Proteins Common Name
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
